Cardiac imaging by random access microscopy
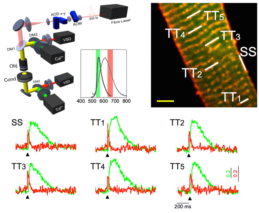
Action potentials (APs), via the transverse axial tubular system (TATS), synchronously trigger uniform calcium release throughout the cardiomyocyte. In heart failure (HF), TATS structural remodeling occurs, leading to asynchronous calcium release across the myocyte and contributing to contractile dysfunction. In our laboratory, we developed an imaging method to simultaneously assess TATS electrical activity and local calcium release. We documented the presence of TATS elements that failed to propagate AP. Sites, where T-tubules fail to conduct AP, show a slower calcium transient compared with regions with electrically coupled elements. It is concluded that TATS electrical remodeling is a major determinant of altered kinetics, amplitude, and homogeneity of calcium release in HF. Recently the system has been implemented to probe and manipulate the electrical dynamics of subcellular membrane domains in optogenetic-modified cardiomyocytes. We demonstrated that TATS is intrinsically excitable and reveals distinct characteristics of self-generated T-tubular action potentials.
Optical control of the whole heart activity
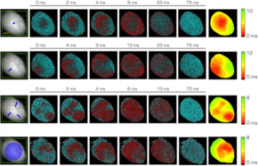
Optogenetics has provided new insights into cardiovascular research, leading to new methods for cardiac pacing, resynchronization therapy, and cardioversion. Although these interventions have clearly demonstrated the feasibility of cardiac manipulation, current optical stimulation strategies do not take into account cardiac wave dynamics in real-time. Here, we developed an all-optical platform complemented by integrated, newly developed software to monitor and control electrical activity in intact mouse hearts. The system combined a wide-field mesoscope with a digital projector for supra- and sub-threshold optogenetic manipulation. Cardiac functionality could be manipulated either in free-run mode with sub-millisecond temporal resolution or in a closed-loop fashion. Recently we found that sub-threshold manipulation also affects the dynamics of cardiac electrical activity, increasing the magnitude of cardiac alternans and we demonstrated that VT stability significantly decreases during sub-threshold illumination primarily due to an increase in the amplitude of electrical oscillations. The development of this innovative optical methodology provides the first proof-of-concept that a real-time optical-based stimulation can control cardiac rhythm in normal and abnormal conditions, promising a new approach for the investigation of the (patho)physiology of the heart.
Whole heart cytoarchitecture at micron-scale resolution
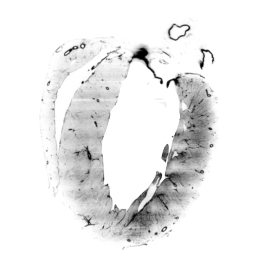
Remodelling processes, associated with genetic and non-genetic cardiac diseases, can cause alterations of the electrical conduction and mechanical dysfunction. Current models employed to predict functional alterations caused by structural remodeling commonly do not draw upon comprehensive functional and structural data and furthermore are often based on low-resolution and non-integrated information. Here, we present a multi-modal optical approach to correlate electrophysiological dysfunction found in a Hypertrophic Cardiomyopathy mouse model with its structural alterations. In detail, we first employed an optical mapping system to characterize action potential propagation in diseased and control whole-heart preparations. To gain the structural data on the same intact hearts, we combined advances in tissue clearing, staining and high-resolution light-sheet microscopy to reconstruct the three-dimensional organization of the cardiac conduction system on a cellular level. In particular, we optimized a passive Clarity protocol for clearing the whole heart and for achieving homogeneously fluorescent probe penetration into the entire tissue. Moreover, we developed a cytoarchitectonic analysis software to identify cells and to map fibers alignment in three-dimensions. The structural reconstruction was directly used to generate a high-resolution image-based computational model to simulate the conduction pathway of action potential propagation across the whole organ with the aim to elucidate the role of cellular disorganization in the electrical dysfunctions mapped previously through optical mapping. We believe that this innovative experimental approach will pave the way for a unifying model which integrates functional and structural data and enable a comprehensive investigation of the morphological causes that lead to electrical alterations after structural remodelling.